|
Ion-ion interactions are seen in precipitation reactions, where a solid forms upon the mixing of two aqueous solutions. As for example: metals such as sodium losses electrons to to become positive ion, whereas non-metal such as chlorine accepts. Ion interactions form the strong bonds that hold ionic solids together. Ionic bonds are bonds formed between ions with opposite charges. Definition: An ionic bond is formed when valence electrons are transferred from one atom to the other to complete the outer electron shell. Ionic bond is a kind of chemical bond which involves an electrostatic attraction between two oppositely charged ions because of the complete transfer of valence electrons between them. Carbon dioxide, water, and chlorine gas are some common examples of compounds having a covalent bond. 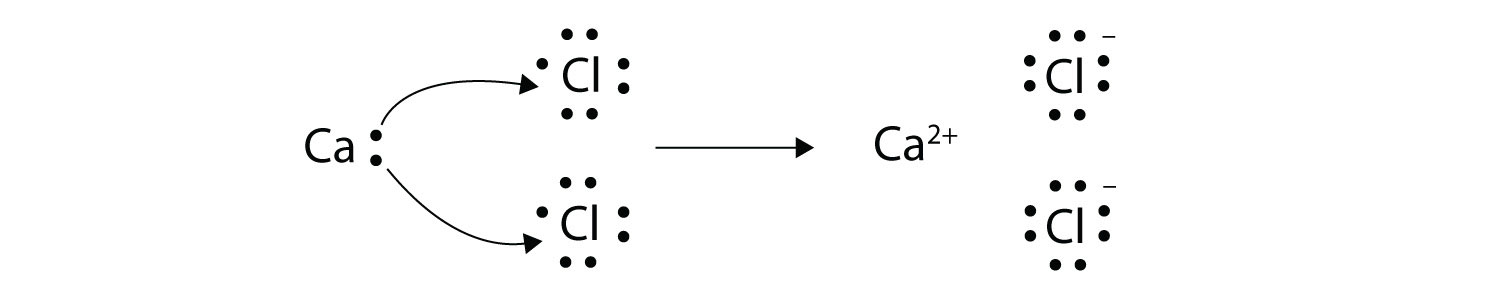
A dipole exists in a molecule which engages in covalent bonding, but one side of the molecule has more electronegativity than the other side. Ions engage in ionic bonding where positive ions (cations) are attracted to negative ions (anions). For example, a saturated solution of sodium chloride contains more ions than the solution can support, and sodium cations will start attracting chloride anions to form solid salt crystals. Technically ions and atoms are categorized differently. There are fairly strong interactions between these ion pairs and free ions, so the clusters grow. They form an ion-pair, a new particle which has a positively-charged area and a negatively-charged area. Eventually, they will stick together and a considerable amount of energy will be required to separate them again. If a vacuum contains two oppositely-charged particles (such as a sodium cation and a chloride anion), they will be attracted to each other, and the force will increase as the two articles approach each other. Bonding energy is generally high, ranging between 6 kJ/mol for example, ionic bond strength for NaCl (i.e., Na Cl ) is about 770 kJ/mol. In general, this bond is nondirectional and has equal strength in all directions. The force depends on the product of the charges \((Z_\)) Hence, cations and anions are bonded together via strong electrostatic attraction, forming the ionic bond. These Coulombic forces operate over relatively long distances in the gas phase. 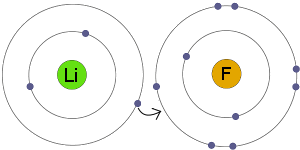
Like charges repel each other and opposite charges attract. They are also referred to as ionic bonds and are the forces that hold together ionic compounds. Ion-ion interactions are an attractive force between ions with opposite charges.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
